Oxaloacetate and PQQ as potent anti lactate agents
-
This post is deleted! -
@user73636 said in Oxaloacetate and PQQ as potent anti lactate agents:
Oxaloacetate
The issue is (as with a lot of other biomolecules), this will instantly get turned into citric acid
-
While a portion of OAA is indeed converted to citrate, this is actually a feature, not a bug, of its anti-lactate profile:
Clearing the Bottleneck: By converting to citrate, OAA "pulls" Acetyl-CoA into the TCA cycle.
Activating PDH: High levels of Acetyl-CoA normally inhibit Pyruvate Dehydrogenase (PDH). When OAA clears that Acetyl-CoA out by turning it into citrate, it "unlocks" PDH.
Result: Pyruvate is now more likely to be converted into Acetyl-CoA (to feed the cycle) rather than being "shunted" into lactate.Cytoplasmic vs. Mitochondrial Activity
The anti-lactate effects described in studies from exogenous OAA often occur in the cytoplasm before the OAA even reaches the mitochondria:
The Redox Buffer: In the cytoplasm, OAA effectively starves the LDH enzyme of its necessary fuel as they both compete for use of nadh
The Malate Shuttle: The resulting Malate then enters the mitochondria, effectively "shuttling" those electrons in for oxidative phosphorylation rather than letting them fester as "reductive stress" in the cytoplasm.Kinetic Inhibition of LDH
OAA is a structural analogue of pyruvate. Because of this similarity, it can act as a competitive inhibitor of Lactate Dehydrogenase (LDH). Even if it eventually gets metabolized, its presence in the cellular pool interferes with the enzyme's ability to bind to pyruvate and create lactate.You are correct that OAA is highly unstable and metabolically "hungry." This is why OAA supplementation often requires higher doses or stabilized forms (like thermally stabilized anhydrous OAA) to ensure enough reaches the target tissues before being decarboxylated or converted.
OAA's instability (spontaneous decarboxylation to pyruvate + CO2, half-life ~minutes at pH 7.4) necessitates stabilized forms like benaGene (anhydrous OAA) at 100-500mg doses for therapeutic levels, ensuring cytoplasmic delivery before mitochondrial uptake or decay. This supports the observed lactate drops in models without rapid citrate exhaustion.
-
Compounds that inhibit lactate overproduction are usually beneficial, but relying on oxaloacetate for this is not ideal.
- Pyruvate carboxylase is required for glutamine-independent growth of tumor cells (PC synthesizes oxaloacetate)
In case of oxaloacetate supplementation, delivery is the first challenge because most of the dose is metabolized in the liver after absorption. For esterified forms, the fraction that reaches target cells is likely overestimated, especially when tissue circulation is poor.
Malate dehydrogenase (MDH) interconverts malate and oxaloacetate, and this enzyme is also part of the malate-aspartate shuttle (MAS) that you mention.
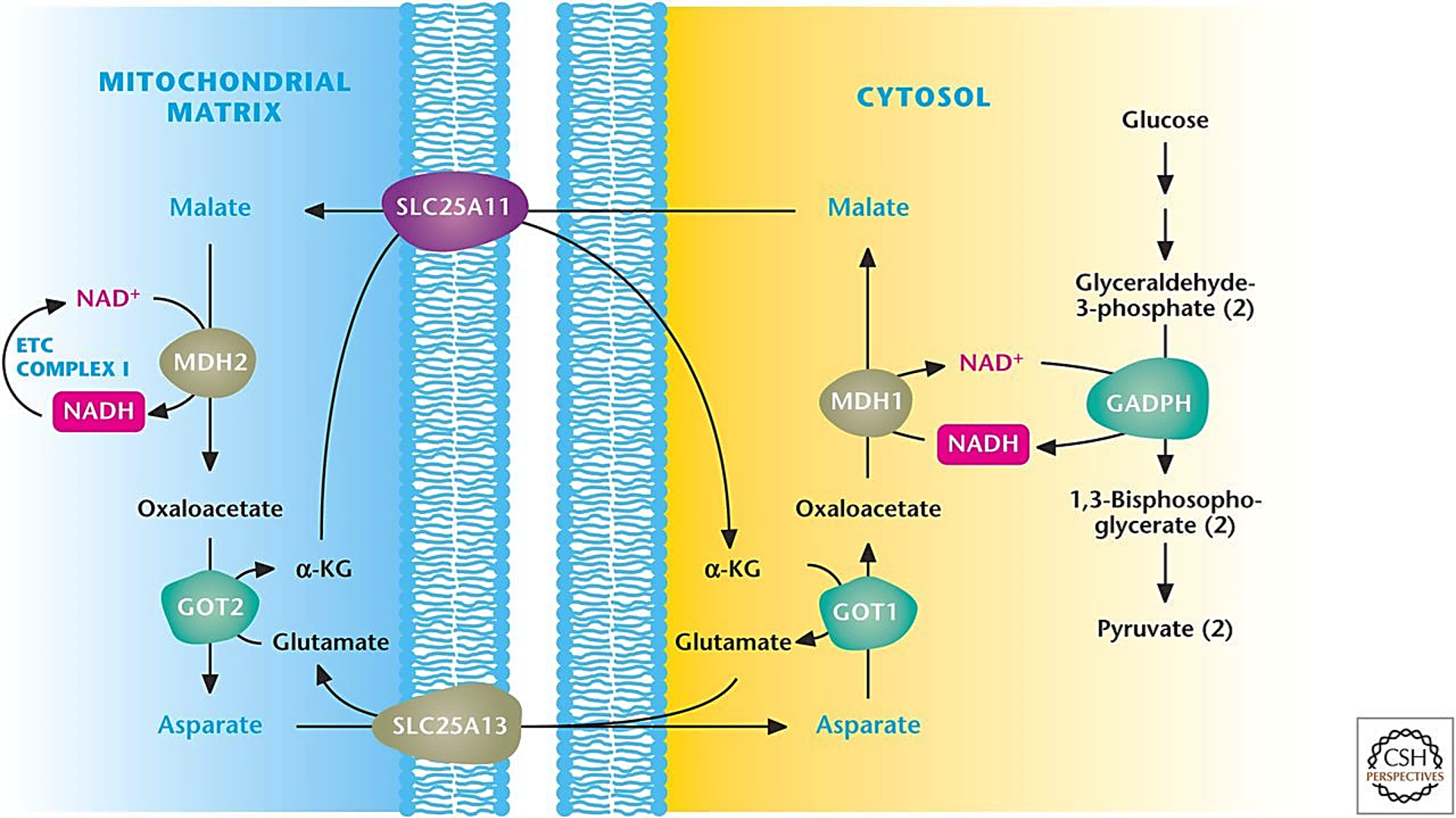
⠀(10.1101/cshperspect.a040543)The figure shows some potential concerns:
Cytosolic NAD reoxidation by MDH can promote glycolysis, but without reliably improving mitochondrial metabolism for further oxidation.
Extra oxaloacetate (OAA) may not convert into malate as expected. Aspartate is another component of the shuttle and is a reaction away from oxaloacetate (GOT/AST is bidirectional).
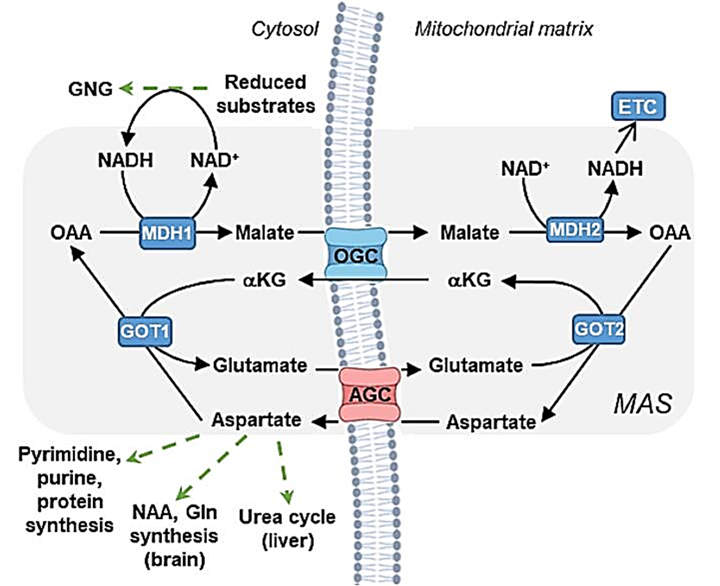
⠀(10.1016/j.ymgmr.2023.100967)- An Essential Role of the Mitochondrial Electron Transport Chain in Cell Proliferation Is to Enable Aspartate Synthesis
- Supporting Aspartate Biosynthesis Is an Essential Function of Respiration in Proliferating Cells
Oxaloacetate enters mitochondria hydrogenated as malate. Recovering oxaloacetate on the mitochondrial side depends on sufficient NAD⁺ (also shown on the figure), which may be scarce and prioritized for other reactions, such as that of KGDHc.
Recovery of oxaloacetate from malate in mitochondria doesn't guarantee its reaction with acetyl-CoA. It may instead support glutamate metabolism by accepting its amino group, yielding ketoglutarate for KGDHc (↻) or IDH (↺). One route doesn't exclude the other, as a fraction of ketoglutarate can undergo oxidative decarboxylation (releasing CO₂) and the other reductive carboxylation (incorporating CO₂),
Supporting glutamate metabolism in forward function:
- Oxaloacetate + glutamate ←{GOT}→ Aspartate + Ketoglutarate
- Ketoglutarate –{KGDHc}→ Succinyl-CoA ←{STK}→ ATP + Succinate
A portion of oxaloacetate-derived malate can also be converted to fumarate in reverse TCA cycle operation.
- Oxaloacetate → Malate → Fumarate → Succinate
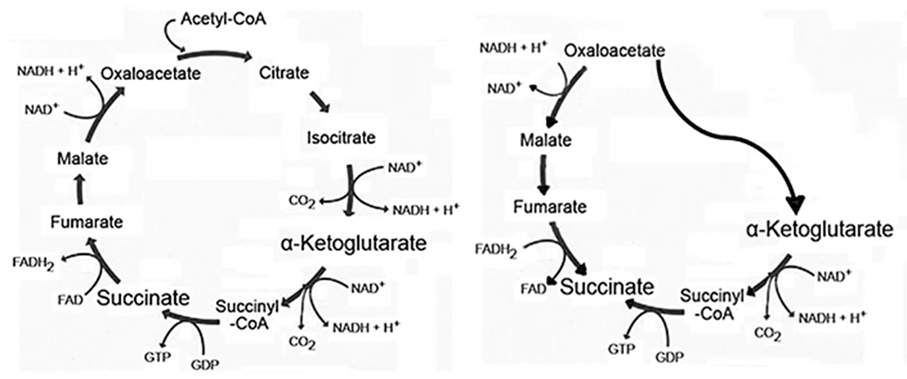
⠀(10.3389/fendo.2012.00022)This way, oxaloacetate helps to generate ketoglutarate to support mitochondrial fermentation and non-respiratory ATP synthesis with minimal oxidation. In addition, fumarate may be used as a substitute to deficient oxygen, accepting electrons from the respiratory chain sourced from ketoglutarate itself, dihydro-orotate, etc.
- (Oxaloacetate +) Glutamate → Ketoglutarate → Succinate ← Fumarate ← Oxaloacetate
Succinate accumulation becomes comparable to lactate. Both metabolites are exported as fermentation end-products together with an extra H⁺, contributing to extracellular acidification.
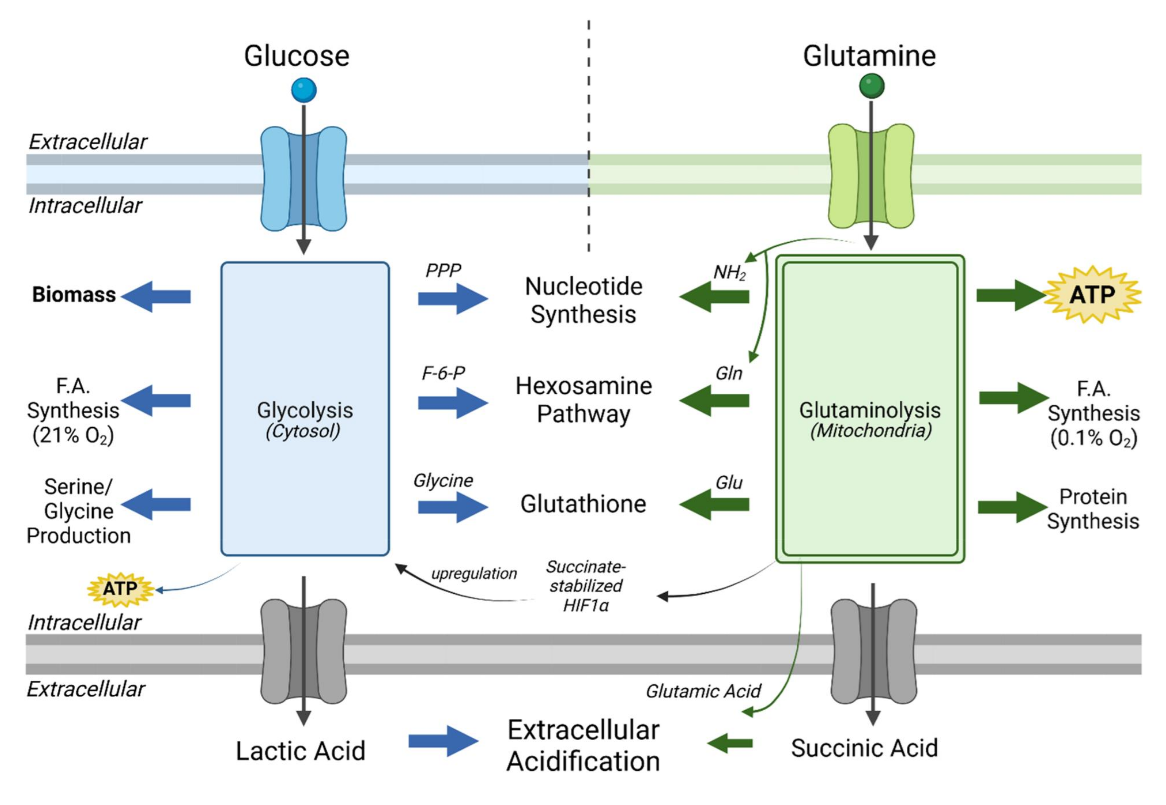
⠀(10.1080/17590914.2024.2422268)"Succinate-stabilized HIF-1a" reinforces PDHc inhibition, giving another reason not to assume that extra oxaloacetate and CoA release will serve PDHc, because they may just as well promote fatty acid oxidation.
If oxaloacetate condenses with acetyl-CoA, that doesn't commit it oxidation either; oxaloacetate can serve as a carrier to export excess acetyl groups to support lipid synthesis.
As for pyruvate and its multiple metabolic fates, even discounting lactate and oxaloacetate in this context, we can't assume that pyruvate routing to the chronically inhibited PDHc will prevail over supporting an upregulated pathway, such as in metabolizing abundant glutamate via GPT/ALT, which remains expressed outside the liver.
- Pyruvate + H⁺ + CoA + NAD⁺ –{
 PDHc}→ Acetyl-CoA + CO₂ + NADH + H⁺
PDHc}→ Acetyl-CoA + CO₂ + NADH + H⁺ - Pyruvate + ⇈Glutamate ←{GPT}→ Alanine + Ketoglutarate
This is another potential route that would supply ketoglutarate without depending on glutamate oxidation, sparing oxidative capacity to keep KGDHc running.
Hello! It looks like you're interested in this conversation, but you don't have an account yet.
Getting fed up of having to scroll through the same posts each visit? When you register for an account, you'll always come back to exactly where you were before, and choose to be notified of new replies (either via email, or push notification). You'll also be able to save bookmarks and upvote posts to show your appreciation to other community members.
With your input, this post could be even better 💗
Register Login