Peat diet and the risk of Vitamin A toxicity, fatty liver
-
Just to remind that Okinawa is a place well known for the number of centenarians and they used to eat a low-fat diet but with 800g/day of sweet potato, at least in the past. What's that, like 250.000 IU beta-carotene? Which is many times more than the carrot that a certain detective is upset about.
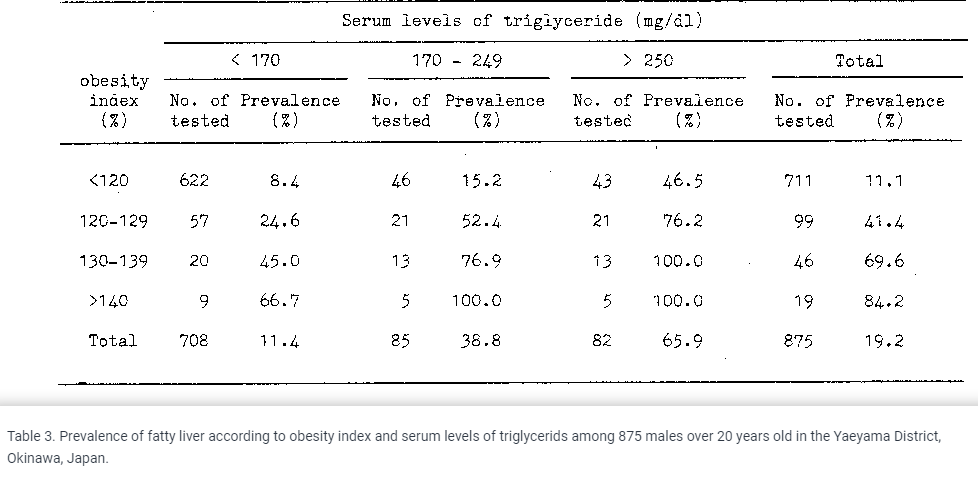
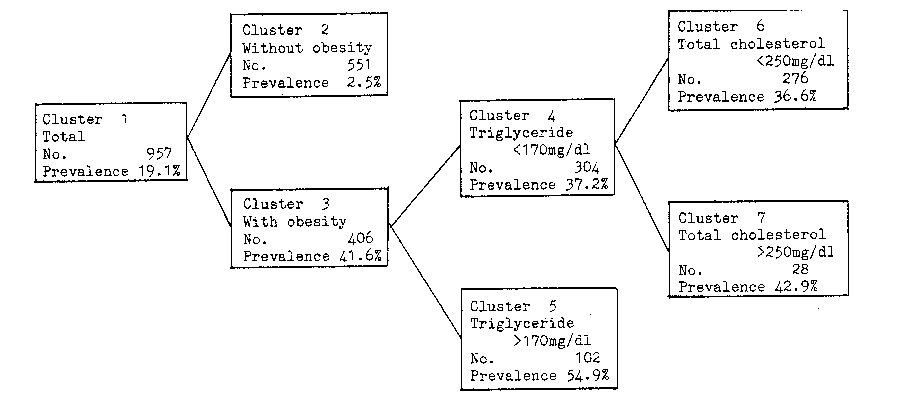
If you're not eating liver every day and you're healthy, I highly doubt that retinol is a problem, or that eating 1 carrot will poison you with beta-carotene haha
-
I supplement vitamin A (100k IU Retinol equivalent from retinyl acetate) for a few days if I start getting dandruff or if I want better skin
-
@GreekDemiGod Vitamin A is not toxic. In fact it could lower and probably has the same antagonistic effect to serotonin as vitamin E does to estrogen.
-
The thyroid hormones associate with three types of simple proteins in the serum: Transthyretin (prealbumin), thyroid binding globulin, and albumin. -Ray Peat, PhD
Because the actions of T3 can be inhibited by many factors, including polyunsaturated fatty acids, reverse T3, and excess thyroxine, the absolute level of T3 can’t be used by itself for diagnosis. “Free T3” or “free T4” is a laboratory concept, and the biological activity of T3 doesn’t necessarily correspond to its “freedom” in the test. T3 bound to its transport proteins can be demonstrated to enter cells, mitochondria, and nuclei. -Ray Peat, PhD
Transthyretin, which carries both vitamin A and thyroid hormones, is sharply decreased by stress, and should probably be regularly measured as part of the thyroid examination. -Ray Peat, PhD
Transthyretin, carrying the thyroid hormone, enters the cell’s mitochondria and nucleus (Azimova, et al., 1984, 1985). In the nucleus, it immediately causes generalized changes in the structure of chromosomes, as if preparing the cell for major adaptive changes. Respiratory activation is immediate in the mitochondria, but as respiration is stimulated, everything in the cell responds, including the genes that support respiratory metabolism. –Ray Peat, PhD
The great difference in water/oil solubility affects the strength of binding between a fatty acid and the lipophilic, oil-like, parts of proteins. When a protein has a region with a high affinity for lipids that contain double bonds, polyunsaturated fatty acids will displace saturated fats, and they can sometimes displace hormones containing multiple double bonds, such as thyroxine and estrogen, from the proteins that have a high specificity for those hormones. Transthyretin (also called prealbumin) is important as a carrier of the thyroid hormone and vitamin A. The unsaturation of vitamin A and of thyroxin allow them to bind firmly with transthyretin and certain other proteins, but the unsaturated fatty acids are able to displace them, with an efficiency that increases with the number of double bonds, from linoleic (with two double bonds) through DHA (with six double bonds).
The large amount of albumin in the blood is important in normal fatty acid binding and transport, but it is also an important part of our detoxifying system, since it can carry absorbed toxins from the intestine, lungs, or skin to the liver, for detoxification. Albumin facilitates the uptake of saturated fatty acids by cells of various types (Paris, et al., 1978), and its ability to bind fatty acids can protect cells to some extent from the unsaturated fatty acids (e.g., Rhoads, et al., 1983). The liver's detoxification system processes some polyunsaturated fats for excretion, along with hormones and environmental toxins…..
Albumin and transthyretin are two proteins that are sometimes found in large quantities inside cells, and their primary importance is that they bind and transport biologically active oily molecules.
While the competition by PUFA for protein binding sites blocks the effects of thyroid hormone and vitamin A, the action of PUFA on the sex steroid binding protein (SBP, or SSBG, for sex steroid binding globulin) increases the activity of estrogen. That's because the SSBG neutralizes estrogen by binding it, keeping it out of cells; free PUFA keep it from binding estrogen (Reed, et al., 1986). People with low SSBG/estrogen ratio have an increased risk of cancer. When the SSBG protein is free of estrogen, it is able to enter cells, and in that estrogen-free state it probably serves a similar protective function, capturing estrogen molecules that enter cells before they can act on other proteins or chromosomes. Transthyretin, the main transporter of thyroid and vitamin A, and albumin (which can also transport thyroid hormone) are both able to enter cells, while loaded with thyroid hormone and vitamin A. Albumin becomes more lipophilic as it binds more lipid molecules, so its tendency to enter cells increases in proportion to its fat burden. Albumin in the urine is a problem associated with diabetes and kidney disease; albumin loaded with fatty acids passes from the blood into the urine more easily than unloaded albumin, and it is the fatty acids, not the albumin, which causes the kidney damage (Kamijo, et al., 2002). It's possible that SSBG's opposite behavior, entering cells only when it carries no hormones, is the result of becoming less lipophilic when it's loaded with estrogen….
https://raypeat.com/articles/articles/fats-functions-malfunctions.shtml
In Alzheimer’s disease, the commonly recognized features are tangles, amyloid deposits, hypometabolism, and evidence of inflammatory processes. Cells related to inflammation can produce amyloid, as well as remove it. Glycation, the attachment of sugar molecules to proteins, can happen quickly, and can occur either with or without enzyme catalysis. The failure of glucose consumption and of carbon dioxide production in Alzheimer’s disease predisposes to glycation.
Glycation imitates mutated forms of proteins, for example normal transthyretin behaves like the prion protein, forming amyloid. Transthyretin, the protein that carries thyroid hormone and vitamin A, is normally taken up along with cholesterol under the influence of thyroid hormone. Abnormal cholesterol metabolism is one of the traits associated with Alzheimer’s disease. In the absence of thyroid-supported respiration, carbon dioxide and other respiration-associated molecules (e.g., acetate) are replaced by lactate and unused sugar, causing abnormal modifications of proteins such as tau, which regulates microtubule assembly. Glycation of collagen in the extracellular matrix alters the properties of the matrix. The glycated matrix would become a preferred site for glycated prion-like proteins.
It is possible that the altered transthyretin makes vitamin A less available to cells. Vitamin A deficiency creates major disruption of the framework proteins. Fragments of starch molecules inhibit the enzymes that remove inappropriately bound sugar molecules from proteins, and the inability to metabolize sugar into carbon dioxide increases that binding. Starches and unsaturated fats cooperate in this process of inappropriate sugar binding, while thyroid hormone, and the carbon dioxide it produces, tend to prevent the binding…
...................
Retinol Binding Protein and Its Interaction with Transthyretin
Transport of vitamin A to the target cells is mediated by the lipocalin retinol-binding protein. In plasma, RBP is found in a complex with its carrier protein Transthyretin (TTR). The structures of RBP free and in complex with TTR provide the details of the protein–protein interaction.
Whether vitamin A is to be ultimately utilized as retinoic acid, 11-cis-retinal or another retinoid, RBP delivers only all-trans-retinol, and only retinol can trigger secretion of RBP.4 In the plasma, **RBP binds to the larger protein, transthyretin (TTR, previously referred to as thyroxine binding prealbumin). The binding of RBP to TTR was suggested to prevent extensive loss of the low molecular weight RBP through glomerular filtration.**3 This hypothesis was supported by the much later experiments of Blaner and colleagues with TTR “knockout” mice that demonstrate that RBP is rapidly clearly from the plasma in TTR deficient mice.11 In vitro one tetramer of TTR can bind two molecules of retinol binding protein. However, the concentration of RBP in the plasma is limiting and as a consequence the complex isolated from serum is composed of TTR and RBP in a one to one stoichiometry with a resultant molecular mass of about 80000 Da.
...
As indicated, complex formation prevents extensive loss of RBP through glomerular filtration and the loss of the two carboxy-terminal leucines (182-183) that are found nestled in a groove formed by the docking of RBP on to TTR may lead to a reduced affinity of RBP for TTR.
A variety of naturally occurring mutations in transthyretin have been described, and the mutation of Ile(84) to Asn or Ser abrogates RBP:TTR complex formation. 26 Individuals with this variant of TTR have substantially lowered plasma concentrations of RBP due to kidney filtration,27 an observation which confirms the importance of complex formation in vitamin A homeostasis.
https://www.ncbi.nlm.nih.gov/books/NBK6223/
Urinary Retinol-Binding Protein: Relationship to Renal Function and Cardiovascular Risk Factors in Chronic Kidney Disease
Serum RBP and urinary RBP showed a positive association. It has been previously proposed that high levels of urinary RBP could be a consequence of high serum RBP levels [21,22]. RBP4, which accounts for almost the total content of circulating RBP, has been recently recognized as an adipocine, and is related to insulin resistance, obesity, diabetes and fat mass [23–26]. Thus, RBP4 could confound the relationship between urinary RBP and renal function or cardiovascular risk factors. In addition, decreasing eGFR is related to increasing serum RBP, a fact that could contribute to this confounding effect. Once adjustments were made, with a particular emphasis on renal function, the positive association between serum and urinary RBP was no longer significant. In addition, the other relationships described in the manuscript were also independent of the effect of serum RBP.
In conclusion, our results suggest that urinary RBP is significantly associated to renal function in general CKD and should be further explored as a predictive and independent marker of CKD progression.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5031461/
Urinary RBP as an Independent Predictor of Renal Outcome in Diabetic Nephropathy
Patients with higher urinary RBP manifested dysregulation of cholesterol metabolism with higher levels of total cholesterol, LDL-C and HDL-C. Increased levels of cholesterol and LDL-C are linked to lipid accumulation and lipotoxicity in tissue. Heavy lipid droplet deposition is observed in renal tubules in DN and associated with tubular damage [18–20]. Uptake of cholesterol is increased by tubular epithelial cells in DN with downregulation of cholesterol efflux genes, e.g., ABCA1, ABCG and upregulation of lipoprotein receptors, e.g., LOX-1, CD36 [18, 21, 22], and aggravates oxidative stress in tubular epithelial cells. In addition, oxidized LDL could directly induce inflammation, apoptosis and fibrosis on tubular epithelial cells [23–25]. And oxidized LDL was reported able to predict eGFR deterioration in proteinuric diabetic kidney disease [23]. In our study, HDL-C levels concurrently increased. HDL-C is recognized as the good cholesterol, which can reverse cholesterol transport from peripheral tissues. Increased HDL-C levels may protect patients from cardiovascular disease [26]. However, recent studies found that in CKD patients, HDL-C had altered structure and became pathogenetic [27, 28]. Dysfunctional HDL-C led to endothelial injury [28], whether they exert deleterious effects on tubular epithelial cells was unclear and requires further study. Altogether, tubular cells are susceptible to lipid disorders. Our study indicated that dysregulated lipid metabolism increased with tubular damage in DN. But the use of statins had a limited effect on the alleviation of lipid disorders. Novel drugs that able to remove ectopic lipid deposition in renal tubules might alleviate tubular damage.
https://www.hindawi.com/journals/dm/2022/9687868/
Transthyretin (TTR) is a tetrameric transport protein highly conserved through vertebrate evolution and synthesized in the liver, choroid plexus, and retinal pigment epithelium. TTR transports the thyroid hormone thyroxine and the retinol-binding protein (RBP) bound to retinol (vitamin A). Mutations in TTR are associated with inherited transthyretin amyloidosis (ATTRv), a progressive, debilitating disease that is ultimately fatal and is characterized by misfolding of TTR and aggregation as amyloid fibrils, predominantly leading to cardiomyopathy or polyneuropathy depending on the particular TTR mutation. Transthyretin amyloid cardiomyopathy can also occur as an age-related disease caused by misfolding of wild-type TTR.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7606379/
Transthyretin is a prealbumin protein of 127 amino acids and is a primary transport protein for thyroxine and retinol (vitamin A).
Transthyretin has two identical thyroid hormone binding sites, but normally only one of them is occupied. The normal serum transthyretin concentration is 250 mg/L (Table 2), corresponding to maximal binding capacity of 2 mg T4/L. Transthyretin binds approximately 10% of T4 and 10% of T3. In addition, transthyretin also binds retinol-binding protein, thereby being involved in vitamin A transport. Transthyretin is mostly of liver origin, but it is also synthesized in pancreatic islet cells, retina, and epithelial cells of choroid plexus in both rats and humans. Transthyretin synthesized in the choroid plexus may be important for brain development by maintaining the appropriate T4 concentration in the central nervous system and favoring its uniform distribution in different areas of the central nervous system (Bartalena, 1990).
https://www.sciencedirect.com/topics/medicine-and-dentistry/transthyretin
...............
Transthyretin-Binding Activity of Complex Mixtures Representing the Composition of Thyroid-Hormone Disrupting Contaminants in House Dust and Human Serum
House dust contains many organic contaminants that can compete with the thyroid hormone (TH) thyroxine (T4) for binding to transthyretin (TTR). How these contaminants work together at levels found in humans and how displacement from TTR in vitro relates to in vivo T4-TTR binding is unknown.
Objectives: Our aims were to determine the TTR-binding potency for contaminant mixtures as found in house dust, maternal serum, and infant serum; to study whether the TTR-binding potency of the mixtures follows the principle of concentration addition; and to extrapolate the in vitro TTR-binding potency to in vivo inhibition levels of T4-TTR binding in maternal and infant serum.
Methods: Twenty-five contaminants were tested for their in vitro capacity to compete for TTR-binding with a fluorescent FITC-T4 probe. Three mixtures were reconstituted proportionally to median concentrations for these chemicals in house dust, maternal serum, or infant serum from Nordic countries. Measured concentration–response curves were compared with concentration–response curves predicted by concentration addition. For each reconstituted serum mixture, its inhibitor–TTR dissociation constant (Ki) was used to estimate inhibition levels of T4-TTR binding in human blood.
**Results: The TTR-binding potency of the mixtures was well predicted by concentration addition. The ∼20% inhibition in FITC-T4 binding observed for the mixtures reflecting median concentrations in maternal and infant serum was extrapolated to 1.3% inhibition of T4-TTR binding in maternal and 1.5% in infant blood. For nontested mixtures reflecting high-end serum concentrations, these estimates were 6.2% and 4.9%, respectively.
Discussion: The relatively low estimated inhibition levels at median exposure levels may explain why no relationship between exposure to TTR-binding compounds and circulating T4 levels in humans has been reported, so far. We hypothesize, however, that 1.3% inhibition of T4-TTR binding may ultimately be decisive for reaching a status of maternal hypothyroidism or hypothyroxinemia associated with impaired neurodevelopment in children.**
https://doi.org/10.1289/EHP5911
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7015555/
A variety of anthropogenic compounds has been found to be capable of disrupting the endocrine systems of organisms, in laboratory studies as well as in wildlife. The most widely described endpoint is estrogenicity, but other hormonal disturbances, e.g., thyroid hormone disruption, are gaining more and more attention. Here, we present a review and chemical characterization, using principal component analysis, of organic compounds that have been tested for their capacity to bind competitively to the thyroid hormone transport protein transthyretin (TTR). The database contains 250 individual compounds and technical mixtures, of which 144 compounds are defined as TTR binders. Almost one third of these compounds (n = 52) were even more potent than the natural hormone thyroxine (T4). The database was used as a tool to assist in the identification of thyroid hormone-disrupting compounds (THDCs) in an effect-directed analysis (EDA) study of a sediment sample. Two compounds could be confirmed to contribute to the detected TTR-binding potency in the sediment sample, i.e., triclosan and nonylphenol technical mixture. They constituted less than 1 % of the TTR-binding potency of the unfractionated extract. The low rate of explained activity may be attributed to the challenges related to identification of unknown contaminants in combination with the limited knowledge about THDCs in general. This study demonstrates the need for databases containing compound-specific toxicological properties. In the framework of EDA, such a database could be used to assist in the identification and confirmation of causative compounds focusing on thyroid hormone disruption.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4498237/
....................
Evidence that 4-Hydroxynonenal Mediates Oxidative Stress-Induced Neuronal Apoptosis
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6573293/
Interaction of the Receptor for Advanced Glycation End Products (RAGE) with Transthyretin Triggers Nuclear Transcription Factor kB (NF-kB) Activation
Oxidative stress in human amyloid deposits from patients with FAP has been examined immunohistochemically, using two markers of lipid peroxidation, antihydroxynonenal, and thiobarbituric acid reactive substances (Ando et al, 1997). Furthermore, the levels of protein carbonyl, a marker of protein modifications by free radicals, were determined in FAP patients and healthy controls (Ando et al, 1997). Antihydroxynonenal antibody reacted with amyloid deposits in all types of amyloid tissues examined, and significantly higher levels of thiobarbituric acid reactive substances and protein carbonyl were found in amyloid rich tissues of FAP patients than in the control subjects. These results suggest that lipid peroxidation, via free radical injury, occurs in amyloid deposits in FAP. Whether these effects are triggered upon binding of fibrils to RAGE remains to be investigated.
We hypothesized that similar to what may happen in Alzheimer's disease, binding of TTR to RAGE would result in altered gene expression and cellular properties, such as induction of the transcription factor NF-kB. The NF-kB transcription factor complex is a pleiotropic activator that participates in the induction of wide variety of genes. The active complex is composed of two subunits, p50 and p65. We demonstrated that TTR fibrils can bind cell surface RAGE and modulate cellular properties, at least in part, through activation of the transcription factor NF-kB. Though soluble TTR is able to induce NF-kB expression, the effect is less than that produced by the same concentration of TTR fibrils, and it is reduced to background levels when the soluble protein is complexed with RBP. In FAP tissues we found activation of NF-kB, based on nuclear localization and up-regulation of p50, when compared to non-FAP control tissues. Such NF-kB activation might contribute to increased expression of macrophage-colony stimulating factor, interleukin-6, hemeoxygenase type 1, and other inflammation-associated molecules. In FAP, further investigation is needed to address this issue.
Although constitutive expression of RAGE was found in all of the control individuals, increased expression of RAGE in FAP tissues was observed. This indicates that this receptor may be relevant to the cellular dysfunction in the disorder. A likely explanation for the apparent juxtaposition of TTR deposits with cells expressing high levels of RAGE is that TTR-RAGE interaction triggers activation of NF-kB, which, via the NF-kB DNA binding elements in the RAGE promoter, up-regulates RAGE expression (Li and Schmidt, 1997). Increased levels of receptor promote further TTR-RAGE interaction, thereby propagating the cytotoxic effects of RAGE.
Although these studies represent only a first step in establishing a link between expression of RAGE and patophysiological changes in FAP, they suggest a mechanism in which engagement of RAGE in the presence of TTR fibrils could result in deleterious consequences for cellular function and organic homeostasis. If this hypothesis is correct, blocking access of TTR to this cell surface binding site might considerably retard progression of cellular dysfunction.
https://www.nature.com/articles/3780116
Clin Chem Lab Med. 2002 Dec;40(12):1344-8.
Transthyretin: its response to malnutrition and stress injury. clinical usefulness and economic implications.
Serum transthyretin is an ideal marker for monitoring patients who are malnourished or have metabolic consequences of acute stress injury because it has a short half-life, it measures the level of metabolic deficit, the response to nutritional metabolic support, and because it is a prognostic indicator. Mounting clinical evidence indicates that the use of transthyretin to assess and monitor a patient’s nutritional status results in improved treatment outcomes and lower overall healthcare costs.
T4- and T3-binding proteins of rat liver were studied. It was found that the external mitochondrial membranes and matrix contain a protein whose electrophoretic mobility is similar to that of thyroxine-binding blood serum prealbumin (TBPA) and which binds either T4 or T3. This protein is precipitated by monospecific antibodies against TBPA. The internal mitochondrial membrane has two proteins able to bind thyroid hormones, one of which is localized in the cathode part of the gel and binds only T3, while the second one capable of binding T4 rather than T3 and possessing the electrophoretic mobility similar to that of TBPA. Radioimmunoprecipitation with monospecific antibodies against TBPA revealed that this protein also the antigenic determinants common with those of TBPA. The in vivo translocation of 125I-TBPA into submitochondrial fractions was studied. The analysis of densitograms of submitochondrial protein fraction showed that both TBPA and hormones are localized in the same protein fractions. Electron microscopic autoradiography demonstrated that 125I-TBPA enters the cytoplasm through the external membrane and is localized on the internal mitochondrial membrane and matrix.
The in vivo translocation of thyroxine-binding blood serum prealbumin (TBPA) was studied. It was found that the TBPA-hormone complex penetrates-through the plasma membrane into the cytoplasm of target cells. Electron microscopic autoradiography revealed that blood serum TBPA is localized in ribosomes of target cells as well as in mitochondria, lipid droplets and Golgi complex. Negligible amounts of the translocated TBPA is localized in lysosomes of the cells insensitive to thyroid hormones (spleen macrophages). Study of T4- and T3-binding proteins from rat liver cytoplasm demonstrated that one of them has the antigenic determinants common with those of TBPA. It was shown autoimmunoradiographically that the structure of TBPA is not altered during its translocation.
Data from determination of molecular weight and competitive displacement suggest that T3 and T4 are bound to the same protein in chromatin. It was shown that the antigenic determinants of T3 and T4 for the chromatin-binding protein coincide with those for blood serum thyroxine-binding prealbumin (TBPA). It was found also that the binding either to T3 and T4 decreases proportionally to the amount of the TBPA removed from the subcellular fractions. It may thus be concluded that blood serum TBPA is responsible for the binding to T3 and T4 as well as for the realization of the hormonal response.
https://www.functionalps.com/blog/2012/06/12/transthyretin-thyroid-and-vitamin-a-carrier-protein/
....................
Transthyretin cardiac amyloidosis: an underdiagnosed cause of heart failure1-3*
*Also known as transthyretin amyloid cardiomyopathy (ATTR-CM).
ATTR cardiac amyloidosis is an underdiagnosed, progressive, and infiltrative disease that is often overlooked as a cause of heart failure1-3
**ATTR cardiac amyloidosis is a type of amyloidosis that results in thickened heart walls due to increasing amyloid fibril deposition.**3ATTR cardiac amyloidosis is a type of amyloidosis that may lead to organ dysfunction and significant mortality3-5,7,8
Amyloidosis is a multisystemic disease caused by the deposition of amyloid fibrils in the heart and other organs over time.3,9 These amyloid fibrils are aggregates of the thyroxine and retinol transporter protein transthyretin (TTR).10
TTR is mainly produced in the liver and is assembled into a tetramer, or 4-leaf clover shape that normally dissociates and reforms.3,10 In ATTR cardiac amyloidosis, mutations and/or aging promote dissociation into monomers, which then misfold and aggregate to form amyloid fibrils.3,10
https://transthyretincardiacamyloidosis.pfizerpro.com/about
Case Report
A 60-year-old male of Laos descent who has been in Australia since the 1980s presented with acute onset of peripheral oedema and weight gain one week after receiving the second dose of mRNA Pfizer-BioNTech COVID-19 vaccine. He had no past medical history and was not taking regular medications, including over the counter remedies.
The laboratory investigation revealed serum creatinine 69umol/L and serum albumin 16g/L. 24-hour urine yielded 5g of protein. Renal biopsy showed Congo red staining of amyloid deposits. The myeloma work-up showed a serum free light chain ratio of 0.78, IgG kappa 3g/L paraprotein and positive urine Bence jones protein. The bone marrow biopsy demonstrated 10% plasma cell aggregates via CD138 and amyloid deposit within extramedullary tissue. He received bortezomib, cyclophosphamide and dexamethasone chemotherapy without haematological response and is currently being worked up for a stem cell transplant.
Conclusions
There are implications of S-protein in COVID-19 disease and vaccine, which are still under research in the literature. Our case highlights the importance of the prompt investigation, including biopsy in patients with life changing symptoms post vaccination.
-
In scrapie and many other degenerative diseases (the amyloidoses), proteins condense into fibrils that tend to keep enlarging, with a variety of very harmful effects. The condensation of the “amyloid” proteins is sensitive to temperature, and a slight increase in the disorder of the water can induce functional proteins to change their conformation so that they spontaneously associate into fibrous masses. In the absence of sufficient carbon dioxide, all proteins are susceptible to structural alteration by the addition of sugars and fats and aldehydes, especially under conditions that favor lipid peroxidation.
The amyloidoses affect different tissues in different ways, but when they occur in the brain, they produce progressive loss of function, with the type of protein forming the fibrils determining the nature of the functional loss. The protein which carries thyroid hormone and vitamin A, transthyretin, can produce nerve and brain amyloid disease, but it can also protect against other amyloid brain diseases; in Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and the “prion diseases” (scrapie, kuru, CJD, BSE, etc.) amyloid particles are formed by different proteins. The transthyretin protein which is binding small molecules resists condensation into the amyloid fibrils, but without its normal vitamin A and thyroid hormone, it can create toxic fibrils. (Raghu, et al., 2002.)Serum amyloid A, which can increase 1000-fold under the influence of proinflammatory cytokines, resulting from irradiation, stress, trauma, or infection, is an activator of phospholipase A2 (PLA2), which releases fatty acids. Some of the neurodegenerative states, including amyloid-prion diseases, involve activated PLA2, as well as increases in the toxic breakdown products of the polyunsaturated fatty acids, such as 4-hydroxynonenal. The quantity of PUFA in the tissues strongly determines the susceptibility of the tissue to injury by radiation and other stresses. But a diet rich in PUFA will produce brain damage even without exceptional stressors, when there aren’t enough antioxidants, such as vitamin E and selenium, in the diet.
Amyloidosis has traditionally been thought of as a condition involving deposits mainly in blood vessels, kidneys, joints and skin and in extracellular spaces in the brain, and the fact that the “amyloid” stained in a certain way led to the idea that it was a single protein. But as more proteins--currently about 20--were identified in amyloid deposits, it was gradually realized that the deposits can be identified inside cells of many different tissues, before the larger, very visible, extracellular deposits are formed.
There is evidence of a steady increase in the death rate from amyloidosis. It kills women at a younger age than men, often at the age of 50 or 60.
Serum amyloid P is called “the female protein” in hamsters, because of its association with estrogen; castrated (or estrogen treated) males also produce large amounts of it, and its excess is associated with the deposition of amyloid (Coe and Ross, 1985). It can bind other amyloid proteins together, accelerating the formation of fibrils, but this function is probably just a variation of a normal function in immunity, tissue repair, and development.
Estrogen increases the inflammation-associated substances such as IL-6, C-reactive protein, and amyloid, and liberates fatty acids, especially the unstable polyunsaturated fatty acids. It also increases fibrinogen and decreases albumin, increasing the leakiness of capillaries. The decrease of albumin increases the concentration of free fatty acids and tryptophan, which would normally be bound to albumin.
In the U.S. and Europe, livestock are fed large amounts of high-protein feeds, and currently these typically contain fish meal and soybeans. The estrogenic materials in soybeans increase the animals’ tendency toward inflammation (with increased serum amyloid).
Officially, BSE appeared because cows were fed slaughter-house waste containing tissues of sheep that had died of scrapie. Scrapie was a nerve disease of sheep, first reported in Iceland in the 18th century. When I was studying the digestive system and nutrition of horses, I learned that it was common for horses in Norway to be fed dried fish during the winter. This abundant food was probably used for sheep, as well as for horses. The extra protein provided by fish meal is still important for sheep in areas where pastures are limited, but it has now become common to use it to increase productivity and growth throughout the lamb, beef, and dairy industries, as well as in most lab chows fed to experimental animals, such as the hamsters used for testing the infectivity of the diseased tissues.
Increased dietary polyunsaturated fatty acids (PUFA) suppress the activity of the ruminal bacteria which are responsible for the hydrogenation-detoxication of PUFA in the animal’s diet. This allows the unstable fats, 98% of which are normally destroyed, to pass into the animals’ tissues and milk.
The polyunsaturated fats in fish are very unstable, and when they get past the bacterial saturases (biohydrogenases) in the rumen that normally protect ruminants from lipid peroxidation, they are likely to cause their toxic effects more quickly than in humans, whose antioxidant systems are highly developed. The toxic effects of polyunsaturated fats involve altered (immunogenic) protein structure, decreased energy metabolism, and many inflammatory effects produced by the prostaglandin-like substances. Marine fish are now so generally polluted with dioxin, that in Japan there is a clear association between the amount of fish in a person’s diet (their body content of EPA and DHA) and the amount of dioxin in their body.
Radiation and many kinds of poisoning cause early peroxidation of those highly unsaturated fats, and the breakdown products accelerate the changes in the folding and chelating behavior of proteins. The accumulation of altered proteins is associated with the degenerative diseases. The role of toxic metals in brain inflammation is well established (e.g., aluminum, lead, mercury: Campbell, et al., 2004; Dave, et al., 1994; Ronnback and Hansson, 1992).
The “prion hypothesis” has the value of weakening the fanaticism of the DNA-genetics doctrine, but it has some problems. There are now several examples in which other degenerative diseases have been transmitted by procedures similar to those used to test the scrapie agent. (e.g., Goudsmit, et al., 1980; Xing, et al., 2001; Cui, et al., 2002.) Experimental controls haven’t been adequate to distinguish between the pure prion and its associated impurities. Gajdusek burned a sample of the infective hamster brain to ash, and found that it still retained “infectivity.” He argued that there was a mineral template that transmitted the toxic conformation to normal proteins. Others have demonstrated that the active structure of the infective agent is maintained by a carbohydrate scaffolding, or that the infectivity is destroyed by the frequency of ultraviolet light that destroys the active lipid of bacterial endotoxin, lipopolysaccharide.
But simply injuring the brain or other organ (by injecting anything) will sometimes activate a series of reactions similar to those seen in aging and the amyloidoses. When a slight trauma leads to a prolonged or expanding disturbance of structure and function, the process isn’t essentially different from transmitting a condition to another individual. The problem is being “transmitted” from the initial injury, recruiting new cells, and passing the disturbed state on to daughter cells in a disturbed form of regeneration. Keloids, hypertrophic scars, are analogous to the dementias in their overgrowth of connective tissue cells: In the aging or injured brain, the glial cells (mainly astrocytes) proliferate, in reparative processes that sometimes become exaggerated and harmful.
When tissue phospholipids contain large amounts of polyunsaturated fatty acids, large amounts of prostaglandins are immediately formed by any injury, including low doses of ionizing radiation. The liberated free fatty acids have many other effects, including the formation of highly reactive aldehydes, which modify DNA, proteins, and other cell components.
Animals which are “deficient” in the polyunsaturated fatty acids have a great resistance to a variety of inflammatory challenges. Their tissues appear to be poor allergens or antigens, since they can be easily grafted onto other animals without rejection. Something related to this can probably be seen in the data of human liver transplants. Women’s livers are subjected to more lipid peroxidation than men’s, because of the effects of estrogen (increasing growth hormone and free fatty acids, and selectively mobilizing the polyunsaturated fatty acids and increasing their oxidation). Liver transplants from middle-aged female donors fail much more often (40 to 45%) than livers from male donors (22 to 25%), and other organs show the same effect. The autoimmune diseases are several times as common in women as in men, suggesting that some tissues become relatively incompatible with their own body, after prolonged exposure to the unstable fatty acids. If we consider the healthy function of the immune system to be the removal or correction of injured tissue, it’s reasonable to view the random interactions of oxidized fats with proteins as exactly the sort of thing our immune system takes care of.
The serum amyloids A and P and the closely related lipoproteins are considered to be important parts of our “innate immunity,” operating in a more general way than the familiar system of specific acquired immunities.
The amyloids and lipoproteins are powerfully responsive to bacterial endotoxin, LPS, and their structural feature that binds it, the “pleated sheet” structure, appears to also be what allows the amyloids to form amorphous deposits and fibrils under some circumstances. Our innate immune system is perfectly competent for handling our normal stress-induced exposures to bacterial endotoxin, but as we accumulate the unstable fats, each exposure to endotoxin creates additional inflammatory stress by liberating stored fats. The brain has a very high concentration of complex fats, and is highly susceptible to the effects of lipid peroxidative stress, which become progressively worse as the unstable fats accumulate during aging.
More than 60 years ago, a vitamin E deficiency was known to cause a brain disease, sometimes associated with sterility and muscular dystrophy. The symptoms of the brain disease were similar to those of “mad cow disease,” and the condition is now usually called “crazy chick disease.” Veterinarians are usually taught that it is caused by a selenium deficiency, but it is actually the result of an excess of PUFA in the diet, and is exacerbated by increased iron or other oxidants, and prevented by increased vitamin E, selenium, or substitution of saturated fats for the unsaturated.
The modification of proteins’ structure by glycosylation is involved in the development of the toxic form of the “prionic” protein, as well as in all the degenerative processes of aging. Until the ability to use sugar is impaired, cells produce enough carbon dioxide to protect proteins against random glycation, but with each exposure to free polyunsaturated fatty acids, the ability to use glucose is damaged. In the dementias, the brain has a greatly reduced ability to use glucose.
One of estrogen’s central effects is to shift metabolism away from the oxidation of glucose, decreasing carbon dioxide production. There is a much higher incidence of Alzheimer’s disease in women, and estrogen exposure exacerbates all of the changes that lead to it, such as shifts in nerve transmitters, increased vascular leakiness, and the increased production of the acute phase proteins.
Everything that is known about the “always fatal” prionic diseases, the diseases of disturbed protein folding, suggests that they can be avoided and even reversed by systematically reversing the processes that amplify inflammation.
People who take aspirin, drink coffee, and use tobacco, have a much lower incidence of Alzheimer’s disease than people who don’t use those things. Caffeine inhibits brain phospholipase, making it neuroprotective in a wide spectrum of conditions. In recent tests, aspirin has been found to prevent the misfolding of the prion protein, and even to reverse the misfolded beta sheet conformation, restoring it to the harmless normal conformation. Nicotine might have a similar effect, preventing deposition of amyloid fibrils and disrupting those already formed (Ono, et al., 2002). Vitamin E, aspirin, progesterone, and nicotine also inhibit phospholipase, which contributes to their antiinflammatory action. Each of the amyloid-forming proteins probably has molecules that interfere with its toxic accumulation.
Thyroid hormone, vitamins A and E, niacinamide (to inhibit systemic lipolysis), magnesium, calcium, progesterone, sugar, saturated fats, and gelatin all contribute in basic ways to prevention of the inflammatory states that eventually lead to the amyloid diseases. The scarcity of degenerative brain disease in high altitude populations is consistent with a protective role for carbon dioxide.
https://raypeat.com/articles/aging/madcow.shtml...................
Studies involving doxycycline and TUDCA
**The combined doxycycline and TUDCA treatment in lowering TTR deposits was tested in different animal models of FAP. In one study, it was found that the two drugs have a synergistic effect and work by lowering both fibrillar and non-fibrillar deposits. The results of this study, published in the Journal of Translational Medicine, indicate that doxycycline acts as a fibril disruptor in vitro and works by removing TTR amyloid deposits. TUDCA works by lowering the amount of deposited non-fibrillar TTR.These positive results led to the investigation of the effect of doxycycline plus TUDCA in patients with transthyretin amyloidosis, or ATTR amyloidosis, of which FAP is a subtype.
The results of a Phase 2, open-label study of doxycycline (100 mg twice daily) plus TUDCA (250 mg three times a day), published in the journal Amyloid, indicated that the combination of the two compounds stabilized the disease in patients. The treatment was well tolerated and halted the progression of heart disease as well as neuropathy in patients for at least one year.**
https://fapnewstoday.com/doxycycline-and-taurodesoxycholic-acid-tudca/ -
@CO3 Vitamin A seems to increase iodine usage and it's entirely possible that increased vitamin A requires increased iodine. Populations that eat lots of sweet potatoes tend to get high iodine as well (and they also live extremely long lives)
-
Lots of nutrients work inefficiently when there is exesses of estrogen and PUFA and eating higher amounts of fiber is a good way to detox estrogen freeing up the liver to help rid the body of PUFAs. More than likely by eating the higher amounts of fiber this detox is happening leading to better usage of these nutrients like A and copper which seems to follow estrogen too. This is one of the reason Peat recommends the carrot salad, mushrooms, bamboo shoots or even oat or wheat bran.
It's weird to think people underestimate the potency of estrogen and pufa as they increase each others effects on the body especially people who know about Ray Peat. If Androgens are low these issues are much worse as there is no defence against this. Looking from a macro perspective is sometimes best as it can be easy to get lost down a road of reduction
-
This is a topic for the retard forum. Why does it have to get dragged into here too? Do you really have to discuss 'vitamin is toxic' in the place that was created (partially) in reaction to the absurd outgrowth of that idea and legion of fools following it?
-
@TexugoDoMel, when we think that they've dodged toxins and escaped misfortune because their sweet potatoes are purple and low in macabrotenoids, the same detective reappears to add that polyphenols are toxins too. I'm not making this up.
-
@Amazoniac rabbit holes galore. makes for many shades and tints between red and blue pilling. how nice. in itself a new field of science is born to supplant virology and quantum physics.
-
@GreekDemiGod said in Peat diet and the risk of Vitamin A toxicity, fatty liver:
i-
greekdemigod u are just as retarded as the people at raypeat forum /thread
-
@raypneat blocked and reported. Get out.
-
@GreekDemiGod serotonic behavior
-
@Hando-Jin @GreekDemiGod can't think and will believe anything charlie says on that forum, also "what is the best idealabs combo?" hurr durrr
Hello! It looks like you're interested in this conversation, but you don't have an account yet.
Getting fed up of having to scroll through the same posts each visit? When you register for an account, you'll always come back to exactly where you were before, and choose to be notified of new replies (either via email, or push notification). You'll also be able to save bookmarks and upvote posts to show your appreciation to other community members.
With your input, this post could be even better 💗
Register Login